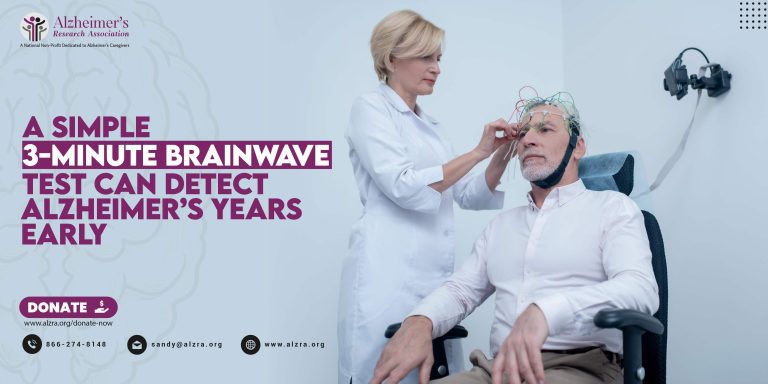
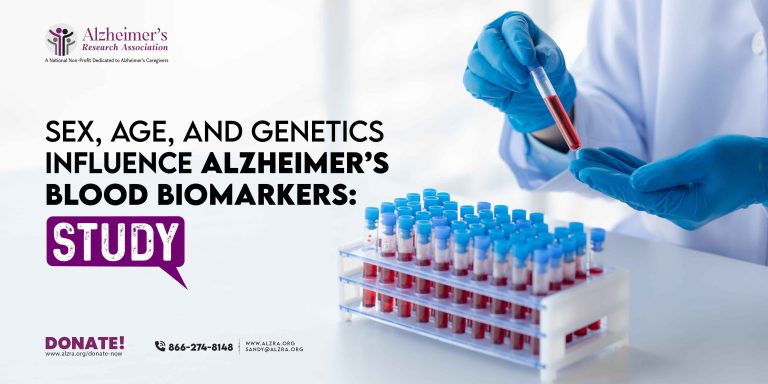
In-Home Senior Care: When Does Your Loved One Need It?
Taking on the role of caregiver for a parent or loved one is a noble but often challenging endeavor. Most seniors want to stay independent, but as daily tasks become harder or health needs increase, subtle signs begin to appear: missed appointments, difficulty keeping up with household chores, or growing concerns about safety. These moments … Continue reading "In-Home Senior Care: When Does Your Loved One Need It?"